
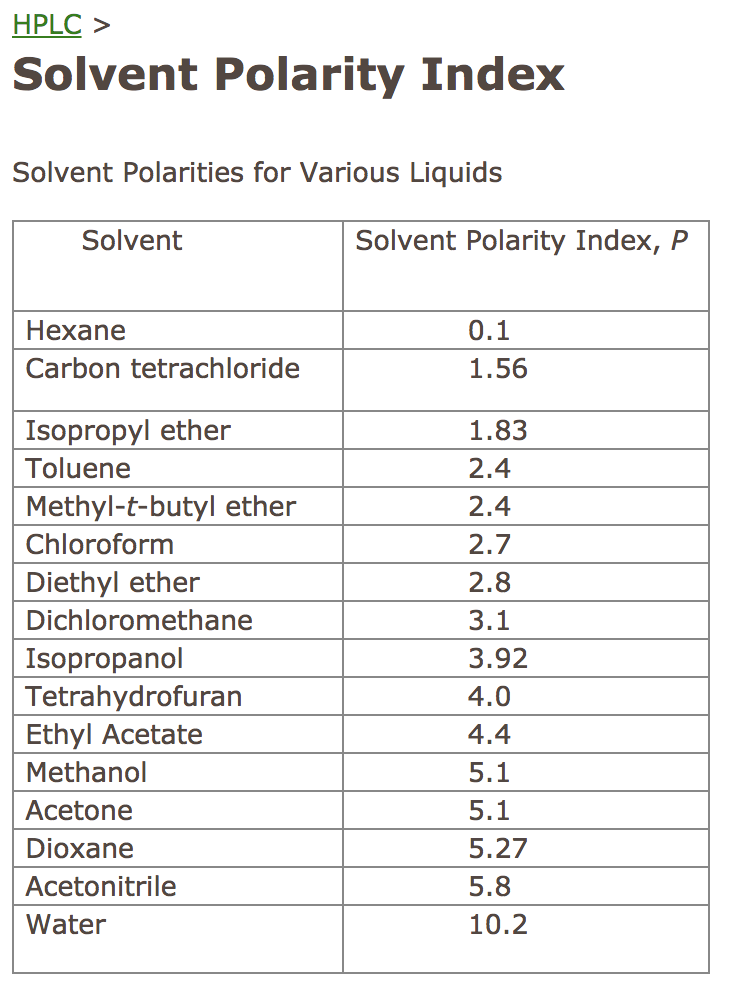
The UV/VIS spectrum of this ester (in contrast to those of five-co-ordinate alkylcobyrinic acid heptamethyl esters) shows a marked solvent dependence, indicating strong interaction between the solvent and the δ+Co–SO 3 δ– dipole, and it is suggested that the main variable which causes the decrease in a and b with solvent polarity is the dipolar solvation term. polarity difference between adsorbent and adsorbate. for any ligand-binding equilibrium in both protic and aprotic solvents. toluene based on polarity in activated carbon fixed-bed reactor. This provides the first direct comparison of the l.f.e.r. The UV activity of toluene can complicate. The results reveal a simple pattern in which the decrease in solvent polarity from water to toluene is accompanied by a decrease in a from 0.36 to 0.25 and in b from –0.3 to –1.7. Polarity indexes of solvents which are commonly used for SEC analysis is shown below. It is one of the only solvent systems where the less polar solvent is typically written first (i.e. dichlorotoluene in an aprotic polar solvent, while distilling off the alcohol. The organic compounds tested are: phenol, nitrophenol, resorcinol, dioxane, acetonitrile and benzotriazole (polar) and toluene, styrene, cyclohexane.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
